A variety of woods occur in nature that is different in color, pattern, smell, etc. It acts as an insulator of heat and electricity and is also known for its acoustic properties. Most students have a question regarding if burning wood is also considered a chemical reaction. So, in this article, we will find out the answer to this question in detail. So, Is the burning of wood a chemical change? Yes, the burning of wood is an example of a chemical reaction called combustion. Here, wood transforms into carbon-di-oxide, water vapor, and ash in presence of heat and oxygen. This process is based upon the pyrolysis of cellulose material that forms wood and also results in the production of a large amount of heat.
The Chemical Reaction of the Burning of Wood
We know that wood is mostly made of cellulose which is actually a carbohydrate i.e. it is formed of carbon, hydrogen, and oxygen atoms. Therefore, the end products formed are also a combination of these atoms rearranged in different patterns. However, there are two types of chemical reactions possible for the combustion of wood. The first chemical equation for the change occurring due to incomplete combustion of wood is as follows: CxHyOz (wood) + O2 + Heat —–> CaHbOc (charred wood) + CO (gas) + Heat This equation actually represents the pyrolysis reaction in which wood is burned incompletely to form charred wood. There is another reaction possible for the burning of wood in which wood is assumed to have burnt completely. The reaction is written below: CxHyOz + O2 + Heat ——> CO2 + H2O + Heat However, both these reactions are hypothetical as multiple side products are formed during the burning of wood. As wood is not a homogeneous material and is available in a number of forms that may be different in their chemical composition, therefore, the products formed due to the burning of wood differ from one sample to another. Other products such as oxides of sulfur, ash, and other volatile organic compounds are also formed during the reaction.
What is a Chemical Change?
The chemical change or chemical reaction is a process in which the atomic arrangement of certain substances, referred to as the reactants, changes under the influence of some external agency such as temperature, pressure, or some other substance. The products formed as a result of a chemical reaction are completely different from the reactants in terms of the atomic arrangement and, therefore, have different chemical and physical properties. To witness a chemical reaction you need not visit a chemistry lab. If you notice your surroundings you can see the chemical reactions happening everywhere around. Making cheese, brewing beer, smelting iron, burning fuels, etc. are a few examples.
The Fundamental concept of Chemical Change
Certain considerations are followed in a chemical reaction. A few of them are given below:
• Conservation of Matter
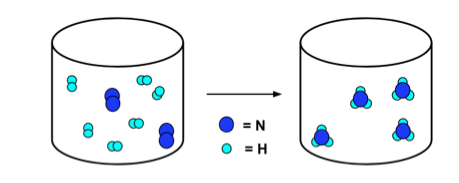
As per the Law of conservation of matter and energy, it can neither be created nor be destroyed but can be transformed from one form to another. This law applies to all chemical reactions and therefore, the chemical equations should always be balanced in terms of atoms. For example, the chemical reaction for the combustion of methane is given as follows: CH4 + 2O2 —-> CO2 + 2H2O In the above reaction to balance the number of hydrogen and oxygen atoms number 2 is placed in front of O2 and H2O as a stoichiometric factor. Stoichiometry refers to the ratio of reactant atoms and product atoms in a chemical reaction equation.
• Kinetic Consideration
The input of energy is mandatory to start any chemical reaction. Like in the case of the burning of wood even when it results in the release of heat an initial spark is required to begin the reaction. Several factors such as catalysts, temperature, the concentration of reactants, etc. influence the rate of reaction.
• Synthesis
A basic characteristic of a chemical reaction is synthesis i.e. one or more products are formed from one or more reactants that are completely different in composition as well as properties.
• Energy Consideration
During a chemical reaction shuffling of atoms take place that requires breaking and formation of chemical bonds. It is known that while breaking the bonds of reactant molecules energy is consumed while when the bonds of the product molecules are formed energy is evolved. However, in certain reactions more amount of energy is consumed, these are known as endoergic reactions while in other reactions more heat is evolved, these are called exoergic reactions. When energy is released in the form of heat the reactions are known as endothermic and exothermic, respectively. Usually, the evolution of heat favors the formation of the product however, entropy is more important in determining the direction of the reaction. The entropy measures the ways in which energy is distributed in a system.
Looking at the chemical change involved in the combustion of wood it can be observed that the law of conservation of matter and energy is followed where the number of reactant atoms is balanced with product atoms. The kinetic considerations as already explained are dependent on the initial source of heat in form of a spark or burning match stick, etc. The combustion of wood results in the synthesis of simpler carbon and hydrogen compounds viz. carbon dioxide, car monoxide, water, etc. Also, although the beginning of the reaction requires a source of heat, still burning wood is an exothermic process in which more heat is released. As all the considerations of a chemical reaction are fulfilled in the process of burning wood, therefore, it is a chemical reaction.
Types of Chemical Change
The chemical reactions are of six types: • Combination Reaction: A single product is formed by the combination of two more reactants. A + B —> AB • Decomposition Reaction: One reactant breaks down to form two or more products. AB —> A + B • Precipitation Reaction: Two solutions combine to form an insoluble product. A + B —-> AB (Precipitate) • Neutralization Reaction: An acid and an alkali react to form a salt. Acid + Base —> Salt • Displacement Reaction: Atoms of one element are replaced by another type of atoms in a compound. AB + C –> AC + B • Combustion Reaction: A compound reacts with oxygen in the presence of heat and gives away carbon dioxide, water vapors, and other gaseous products. This is an exothermic process. A + O2 + Heat —> H2O + CO2 We already know that the burning of wood is a combustion reaction.
Is Burning Wood a Physical Change?
Physical change is the process, in which the composition of a substance remains unaltered, however, rearrangement of molecules does take place. It is temporary in nature and affects only the physical properties of a substance. Usually, physical changes involve the evolution of energy with the least consumption of it. It is a reversible process, for example, freezing or boiling of water, melting of wax, etc. The burning of wood is not a physical process as it involves a change in the chemical composition of wood which forms CO2 and H2O along with other by-products at the end of the reaction. Moreover, it causes a permanent change in the substance and is an irreversible process. Hence, the burning of wood can be correctly called a chemical change.
What is Wood Made of?
Although the composition of wood differs from one species to another there are few components that are universal, however, their percentage also varies between the two species. The main component of wood is cellulose which is a carbohydrate, substance that is made of carbon, hydrogen and, oxygen atoms combined together. The percentage of cellulose components differs from 40-45%. Other components of wood are hemicelluloses which are present in the form of Glucomannan, Glucuronoxylan, and other polysaccharides. Lignin forms the third most abundant component of wood. Extractives present in the wood are low molecular weight compounds that are present in extracellular spaces. Two types of wood occur in nature viz. hardwood and softwood. The main difference between them is their lignin constituent. The lignin in hardwood is a derivative of sinapyl alcohol and coniferyl alcohol while in softwood it is derived from coniferyl alcohol.
Properties of Wood
Wood occurs in nature in a number of varieties which are also different in their properties viz. color, odor, texture, taste, etc. These properties play important role in the identification of different species. The properties such as density and specific gravity also differ from one species to another. Few other properties are given below: • Hygroscopicity: Wood has the tendency to absorb moisture from its surroundings. Although it also absorbs other liquids and gases from the environment, water is most important is due to this property any wood piece always has some amount of moisture. • Swelling & Shrinkage: Wood changes its diameter in accordance with its moisture content. Swelling may occur due to intake of moisture while loss of moisture can result in shrinkage of the wood. • Strength: It also varies drastically among different species which are therefore used for different purposes. • Thermal properties: Wood has a very low thermal conductivity i.e. it does not transfer much heat. However, the thermal conductivity of the wood increases with the moisture content and density of the wood. • Electrical properties: Wood is an insulator. However, if the moisture content is very high it might transmit some amount of electricity. • Acoustic Properties: Wood is known to produce different sounds. Also, it can absorb and amplify sound waves.
Uses of Wood
Various uses of wood are listed as under: • It is used in construction for homes and, fencing and decoration of gardens. • It is used in making tools and kitchen utensils. • It is used for making structures, statues, carvings, etc. • Many musical instruments such as flute, guitar, piano, etc. are made of wood. • It is used in making sports equipment such as cricket bats, hockey sticks, etc. • It is also used for making toys for children. • Commercially wood is used in making furniture, shipbuilding, as fuel, in stationery products, etc.
Conclusion
The burning of wood is a chemical reaction in which complex material is broken into simpler material. It is a form of combustion reaction. Two chemical reactions are possible for the burning of wood, for incomplete combustion the equation is as follows: CXHYOZ + O2 + Heat —> CAHBOC + CO + Heat For complete combustion: CXHYOZ + O2 + Heat —> CO2 + H2O + Heat Fundamental concepts of a chemical reaction include conservation of matter, kinetic consideration, synthesis, and energy considerations.
/184391933-56a34a305f9b58b7d0d14e45.jpg)